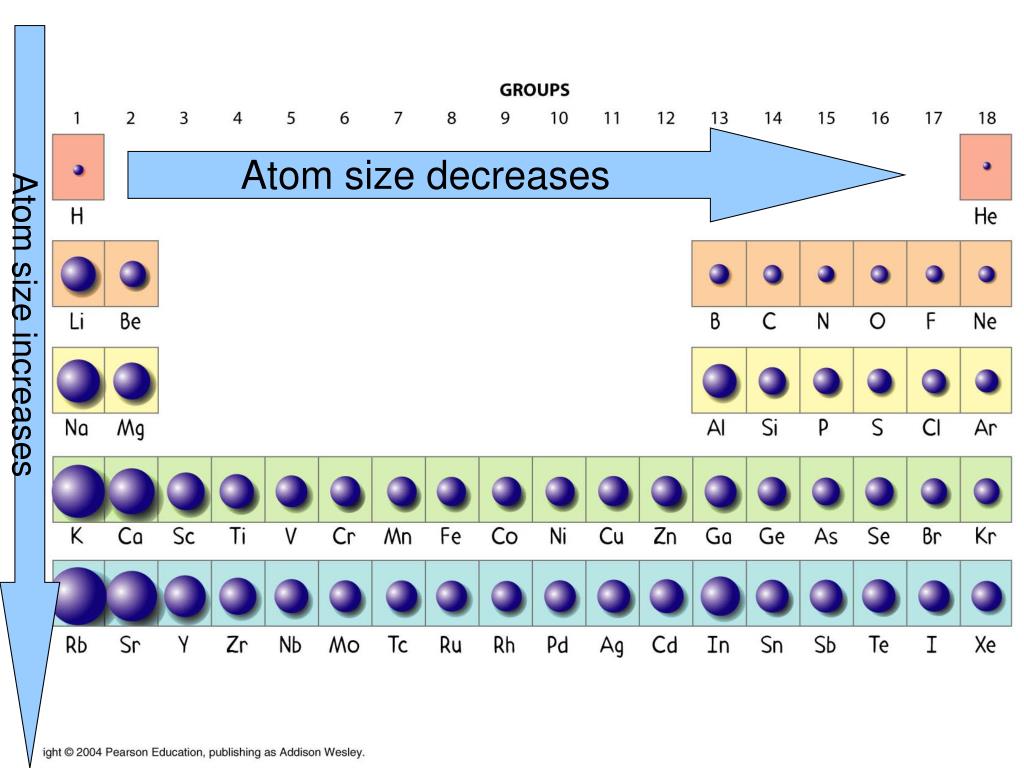
The atomic radii generally decrease because of increase in the number of protons, which is having an attractive effect on the outer mostĮlectrons present in the atom. As the atomic number increases and the number of electrons, the number of shells accommodating those electrons also increases, which means that it will cover more area around the nucleus that results in an increase inĪs one moves along the period, the numbers of electrons increases but the electrons are being added in the same electron shell. The number of electrons will decide the spread of electron cloud around the nucleus of an atom. In neutral atoms, the number of protons and the number of electrons is equal. In the periodic table, the elements are arranged according to their atomic numbers, which is the number of protons present in the elements. The atomic radius of an element cannot be determined accurately because in case where the atoms are combined together, the electron clouds of different atoms overlap which do not give the exact boundary of a single atom. There are various experimental and theoretical methods by which the atomic radii can be calculated. In the above-mentioned diagram, covalent radius is shown by overlapping of two atoms and is less than the sum of two radii, metallic radius forms by just touching of boundaries of the atoms and is exactly equal to the sum of the two radii.In vander waals radis, the atoms remain at some distance and the radius is more than the sun of the two radii. Based on this, atomic radii can be categorized as van der waals radius, ionic radius and covalent radius. The atomic radius is defined on the basis of the type of bonding in which the atom is involved. These atomic dimensions are very small and cannot be seen with the use of an optical microscope. Atomic radius also varies when the atom is subjected to some external force rather than in free space or vacuum. The atomic radius of a free atom varies from that in an atom, which is present in a chemical compound or bound to other atoms by a chemical bond. There are various factors that affect the atomic radius of an atom. Two atoms joined together to form a molecule can be used to find out the atomic radius of an atom. These atomic radii are absolute or more accurate when in vacuum or free space. This electron cloud starts from the nucleus and extends outwards in order to cover a spherical region around it. The space in which the orbits of an atom are present along with the electrons in it is defined as the electron cloud. The size of the atom is given in terms of atomic radii.Īs the shape and size of an atom are not well defined, it is assumed to be spherical in shape and the size of the atom is given by atomic radius. This shape and boundary just gives a rough idea and not well defined. Different orbits are present in different planes and, occupy a significant volume in the 3-D space. These collectively give a spherical shape to the atom of any element. Orbits, in which electrons are present are called electron shells, are not in a single plane. Protons are positively charged species, electrons are negatively charged species, and neutrons are neutral.Īs the electrons are mainly accommodated around the nucleus in orbits that are circular in shape, it occupies some space around the nucleus that decides the size of an atom. The electrons are present in the circular orbits around the nucleus. Neutrons and protons are known as nucleons. The size of an atom is very small and cannot be seen with the naked eyes.A Atoms mainly consist of nucleus that has neutrons and protons in it and electrons are present around the nucleus. An element consists of the same type of atoms and any compound consists of different atoms of different elements. An atom is the smallest unit of any matter that is mainly made of chemical elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
